 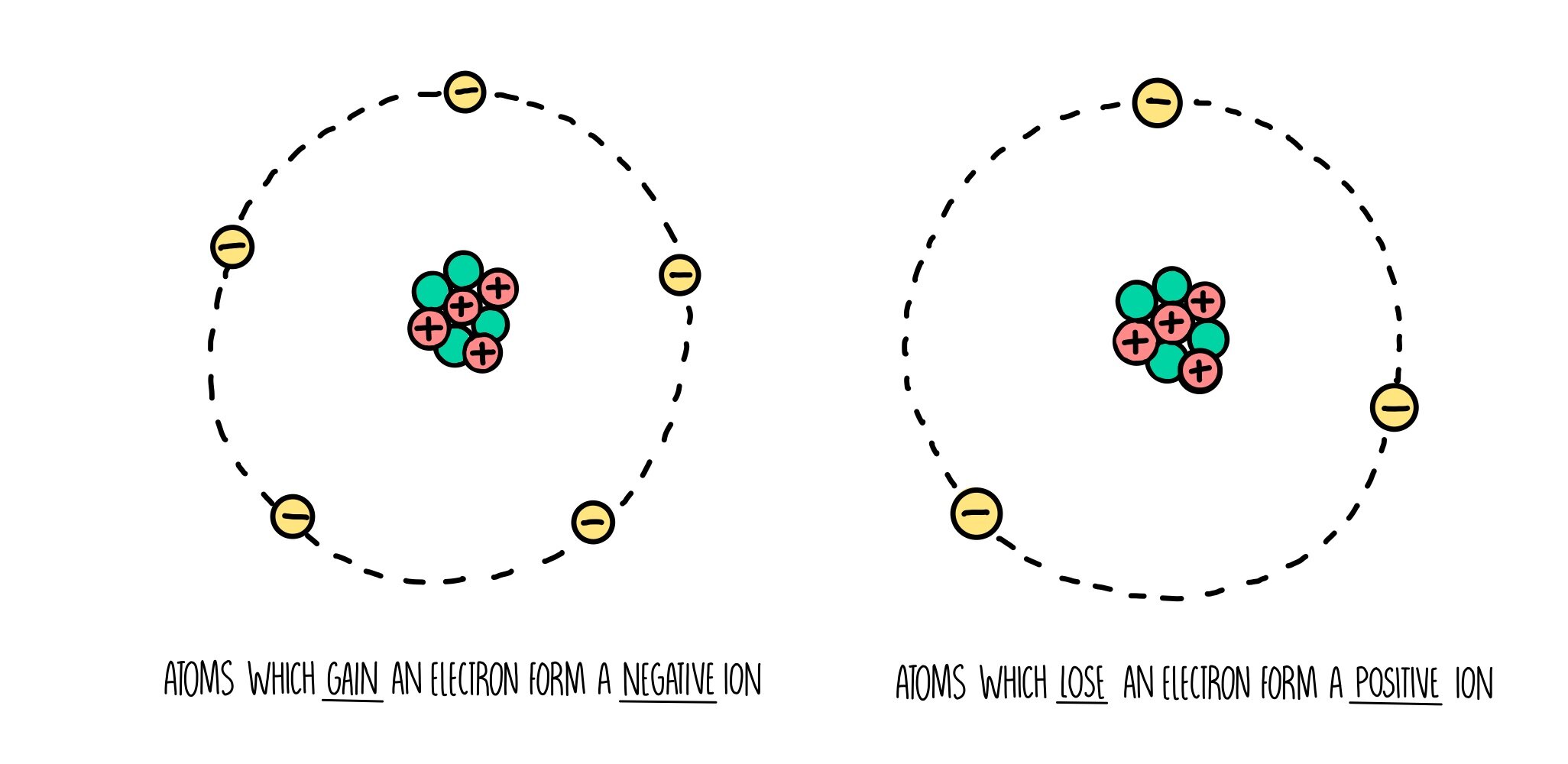
A molecule comprises a group of atoms strongly bound together, and only weakly bonded (if at all) to other molecules. FALSE: there are no molecules in sodium chloride, just ions.(There would also be a fifth sodium ion above the chlorine ion and one more below - but these are not shown in the diagram.) FALSE: In the diagram each chloride ion is attracted to up to four sodium ions by a bond that is an electrostatic force.FALSE: The reason a bond is formed between chloride ions and sodium ions is because they have opposite electrostatic charges - negative and positive.In NaCl there will be six chloride ions strongly bonded to each sodium ion. FALSE: a sodium ion can strongly bond to as many chloride ions as can effectively pack around it in the regular crystal lattice.It is irrelevant how the ions came to be charged. 
FALSE: each positive sodium ion is bonded to each of the neighbouring negative chloride counter-ions.It does not matter how the objects acquired the charge, the attraction only depends on the amount of charge, and the distance between the two charged objects. TRUE: any positively charged object will be attracted to any negatively charged object. 
Following each is a brief comment suggesting whether or not the statement is true, and why. 
There are no molecules shown in the diagram.īelow you will find listed the 20 statements you were asked to think about.A negative ion can be bonded to any neighbouring positive ions if it is close enough.An ionic bond is when one atom donates an electron to another atom, so that they both have full outer shells.A positive ion can only be attracted to one negative ion.In the diagram a sodium ion is attracted to one chloride ion by a bond and is attracted to other chloride ions just by forces.The reason a bond is formed between chloride ions and sodium ions is because they have opposite charges.A negative ion can only be attracted to one positive ion.There is a bond between the ions in each molecule, but no bonds between the molecules.A chlorine atom can only form one ionic bond, because it can only accept one more electron into its outer shell.A chloride ion is only bonded to the sodium ion it accepted an electron from.It is not possible to point to where the ionic bonds are, unless you know which chloride ions accepted electrons from which sodium ions.A negative ion can be attracted to any positive ion.A positive ion can be bonded to any neighbouring negative ions, if it is close enough.An ionic bond is the attraction between a positive ion and a negative ion.In the diagram each molecule of sodium chloride contains one sodium ion and one chloride ion.In the diagram a chloride ion is attracted to one sodium ion by a bond and is attracted to other sodium ions just by forces.The reason a bond is formed between chloride ions and sodium ions is because an electron has been transferred between them.A sodium atom can only form one ionic bond, because it only has one electron in its outer shell to donate.A sodium ion is only bonded to the chloride ion it donated its electron to.A positive ion will be attracted to any negative ion.Please read each statement carefully, and decide whether it is correct or not. RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
